Metals And Non-Metal: This article provides NCERT Solutions for Class 10 Chapter 3 Metals and Non metals. Having a thorough understanding of theories, adequate practice of reactions, equations, and formulas, as well as solving questions from NCERT Chemistry books. Going through the step-wise solutions for each question is crucial for achieving a good score in the board exam.
NCERT Class 10 Science Chapter 3 Notes for Metals and Non metals are provided by the best Chemistry teachers in India. You will also find the step-wise explanation for every question in this article. Going through this step-by-step explanation will help you to understand how to solve the problems better. Continue reading to know NCERT solutions for class 10 science chapter 3 exercise and extra questions and New NCERT Book Class 10 Download Rationalised Chapters PDF.
NCERT Solutions for Class 10 Science Chapter 3 Textbooks Questions
Which of the following pairs will give displacement reactions?
(a) NaCl solution and copper metal.
(b) MgCl2 solution and aluminum metal.
(c) FeSO4 solution and silver metal.
(d) AgNO3 solution and copper metal.
Answer:
(d) AgNO3 solution and copper metal.
rect: Cu(s) + 2AgNO3(aq) → Cu(NO3)2(aq) + 2Ag(s)
Question 2.
Which of the following methods is suitable for preventing an iron frying pan from rusting?
(a) Applying grease
(b) Applying paint.
(c) Applying a coating of zinc
(d) All the above.
Answer:
(c) Applying a coating of zinc.
Question 3.
An element reacts with oxygen to give a compound with a high melting point. This compound is also soluble in water. The element is likely to be
(a) calcium (Ca)
(b) carbon (C)
(c) silicon (Si)
(d) iron (Fe)
Answer:
(a) Calcium. (Ca)
Question 4.
Food cans are coated with tin and not with zinc because
(a) zinc is costlier than tin
(b) zinc has a higher melting point than tin
(c) zinc is less reactive than tin
(d) zinc is more reactive than tin.
Answer:
(d) Zinc is more reactive than tin.
Question 5.
You are given a hammer, a battery, a bulb, wires and a switch.
(a) How could you use them to distinguish between samples of Metals and Non metals?
(b) Assess the usefulness of these tests in distinguishing between Metals and Non metals.
Answer:
(A)
Electrical Conductivity: Metals: Most metals are good conductors of electricity. You can test the electrical conductivity of a sample to determine if it’s a metal. If it conducts electricity, it’s likely a metal.
Non-Metals: Non-metals generally do not conduct electricity, except for some exceptions like graphite (a form of carbon) that can conduct electricity.
Malleability and Ductility: Metals are typically malleable (can be hammered into thin sheets) and ductile (can be drawn into wires). Testing the ability to shape or stretch a sample can indicate whether it’s a metal.
Non-Metals: Non-metals are typically brittle and not malleable or ductile.
Reaction with Acids: Most metals react with acids to produce hydrogen gas. For example, if a sample reacts with hydrochloric acid, it’s likely a metal.
Non-metals: Non-metals do not typically react with acids to produce hydrogen gas.
(B)
Electrical Conductivity: This is a reliable test, as most metals are good conductors. However, it may not work for all non-metals, as some can conduct electricity (e.g., graphite and certain semiconductors).
Malleability and Ductility: While these properties are characteristic of metals, not all metals are equally malleable or ductile. Some non-metals may also exhibit limited malleability (e.g., some metalloids).
Reaction with Acids: This test can be useful for identifying metals that react with acids, but not all metals react with all types of acids. Additionally, some non-metals can react with certain acids to produce gases.
Question 6.
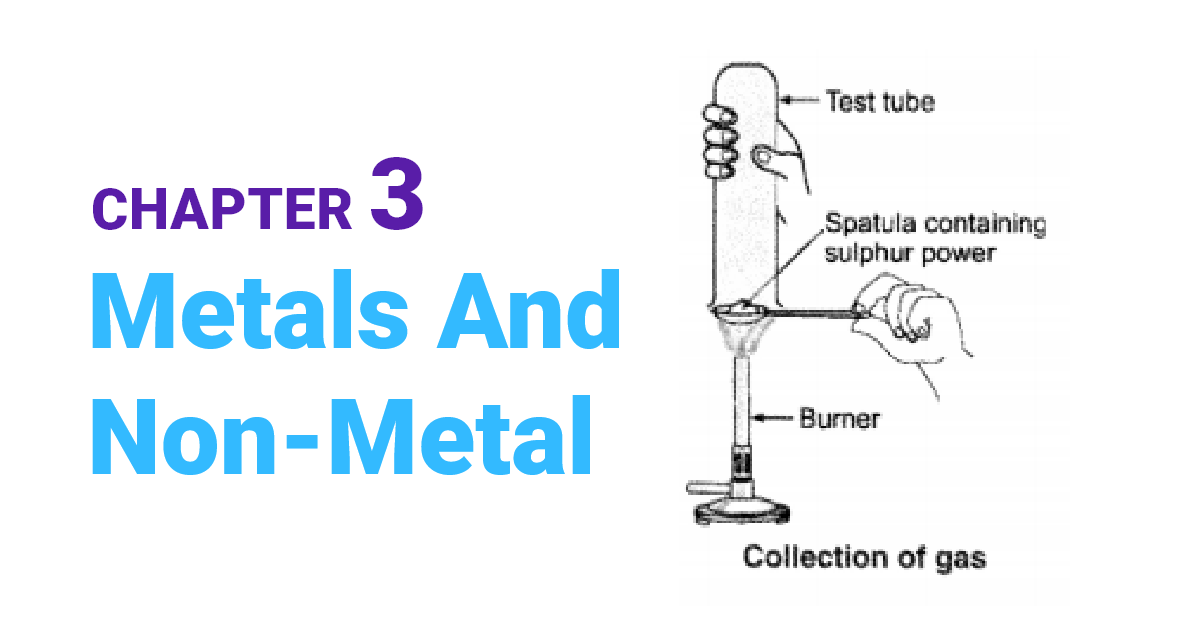
What are amphoteric oxides? Give two examples of amphoteric oxides.
OR Write chemical equations that show aluminium oxide reacts with acid as well as base.
Answer:
Metal oxides that can act as both bases and acids are called amphoteric oxides. In simpler terms, amphoteric oxides are metal oxides that can react with both acids and bases to produce salt and water. Examples of amphoteric oxides include aluminum oxide and zinc oxide.
Question 7.
Name two metals which will displace hydrogen from dilute acids and two metals which will not.
Answer:
(a) Two metals that will displace hydrogen from dilute acids are zinc (Zn) and aluminum (Al).
Zn(s) + 2HCl(aq) → ZnCl2(aq) + H2(g)
2Al(s) + 6HCl(aq) → 2AlCl3(aq) + 3H2(g)
(b) Two metals that will not displace hydrogen from dilute acids are copper (Cu) and silver (Ag).
Question 8.
In the electrolytic refining of a metal M, what would you take as the anode, the cathode and the electrolyte?
Answer:
Cathode – Pure metal
Anode – Impure metal
Electrolyte – Metal salt solution
Question 9.
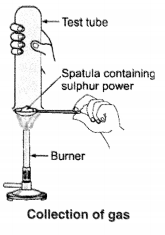
Pratyush took sulphur powder on a spatula and heated it. He collected the gas evolved by inverting a test tube over it, as shown in the figure.
(a) What will be the action of gas on
(i) dry litmus paper?
(ii) moist litmus paper?
(b) Write a balanced chemical equation for the reaction taking place.
Answer:
(A)
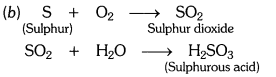
(i) There will be no action on dry litmus paper
(ii)Since the gas is sulphur dioxide (SO2), it turns moist blue litmus paper to red because sulphur
dioxide reacts with moisture to form sulphurous acid.
Question 10.
State two ways to prevent the rusting of iron.
Answer:
Ways to prevent rusting of iron are :
(a) By painting
(b) By galvanizing
Question 11.
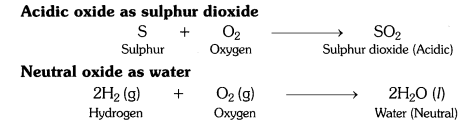
What type of oxides are formed when non-metals combine with oxygen ?
Answer:
Non-metals combine with oxygen to form acidic oxides or neutral oxides.
Question 12.
Give reasons :
(a) Platinum, gold and silver are used to make jewellery.
(b) Sodium, potassium and lithium are stored under oil.
(c) Aluminium is a highly reactive metal, yet it is used to make utensils for cooking.
(d) Carbonate and sulphide ores are usually converted into oxides during the process of extraction.
Answer:
(a) Platinum, gold and silver are used to make jewellery because these are malleable and ductile. These are highly resistant to corrosion.
(b) Sodium, potassium and lithium are very reactive and catch fire when exposed to air. This is due to their low ignition temperature and high reactivity.
(c) Aluminium forms a non-reactive layer of aluminium oxide on its surface. This layer prevents aluminium to react with other substances. That’s why aluminium is used to make cooking utensils.
(d) It is easier to reduce a metal oxide into free metal. Since it is easier to obtain metals from their oxides than from their carbonates or sulphides directly, therefore, the carbonate and sulphide ores are first converted to oxides for extracting the metals.
Question 13.
You must have seen tarnished copper vessels being cleaned with lemon or tamarind juice. Explain why these sour substances are effective in cleaning the vessels.
Answer:
The sour substances such as lemon or tamarind juice contain acids. These acids dissolve the coating of copper oxide or basic copper carbonate present on the surface of tarnished copper vessels and make them shine red-brown again.
Question 14.
Differentiate between metal and non-metal on the basis of their chemical properties.
Answer:
Difference between metals and non-metals
| Metals | Non-metals |
| (i) Metals form basic oxides or amphoteric oxides. | (i) Non-metals form acidic or neutral oxides. |
| (ii) Metals can take the place of hydrogen in acids to create salts. | (ii) Non-metals do not replace hydrogen from acids. |
| (iii) With chlorine, metals make chlorides which are electrovalent. | (iii) With chlorine, non-metals form chlorides which are covalent. |
| (iv) With hydrogen, few metals form electrovalent hydrides. | (iv) With hydrogen, non-metals form many stable hydrides which are covalent. |
Question 15.
A man went door-to-door posing as a goldsmith. He promised to bring back the glitter of old and dull gold ornaments. An unsuspecting lady gave a set of gold bangles to him which he dipped in a particular solution. The bangles sparkled like new but their weight was reduced drastically. The lady was upset but after a futile argument, the man beat a hasty repeat. Can you play the detective to find out the nature of the solution he has used?
Answer:
The dishonest goldsmith dyes the gold bangles with Aqua-regia, which is composed of 1 part nitric acid dissolved in 3 parts hydrochloric acid by volume. This dissolved a significant amount of gold from the gold bangles, resulting in a significant decrease in their weight. A suitable treatment can be used to recover the dissolved gold.
Question 16.
Give reasons why copper is used to make hot water tanks and not steel (an alloy of iron).
Answer:
(i) Copper is a better conductor of heat than steel.
(ii) Copper does not corrode easily. But steel corrodes easily.
(iii) Copper does not react with water at any temperature, whereas iron reacts with water on heating.
NCERT Solutions for Class 10 Science Chapter 3 Index Questions
Page No: 40
- Examples of metals with different properties:
(i) A metal that is a liquid at room temperature is Mercury (Hg). It is the only metal that is liquid at standard conditions and has a relatively low melting point.
(ii) A metal that can be easily cut with a knife is Sodium (Na). Sodium is a soft metal and can be cut with a knife, though it should be handled with care due to its extreme reactivity with moisture and air.
(iii) The metal that is the best conductor of heat is Silver (Ag). Silver is an excellent conductor of both heat and electricity, which is why it’s used in various applications, including electrical contacts and thermal conductors.
(iv) A metal that is a poor conductor of heat is Lead (Pb). Lead has low thermal conductivity, making it a relatively poor conductor of heat. This property is why lead is used as a shield in applications where heat or radiation needs to be contained, such as in certain types of radiation protection. - Meanings of Malleable and Ductile:
- Malleable: Malleability is the property of a material, typically a metal, that allows it to be hammered, rolled, or pressed into thin sheets without breaking or shattering. A malleable material can be deformed under compression and can be shaped into various forms. For example, gold and aluminum are highly malleable and can be easily flattened into thin sheets or foils.
- Ductile: Ductility is the property of a material, usually a metal, that allows it to be stretched or drawn into a thin wire without breaking. Ductile materials can undergo significant deformation under tension and be drawn into wires or threads. For example, copper and iron are ductile metals and can be drawn into wires that are used in electrical cables or construction materials.
Page No: 46
- Why is sodium kept immersed in kerosene oil?
Sodium is kept immersed in kerosene oil to protect it from exposure to oxygen and moisture in the air. Sodium is a highly reactive metal and readily reacts with oxygen and water. When sodium comes into contact with air, it can form a layer of sodium oxide (Na2O) and sodium hydroxide (NaOH) due to its reaction with oxygen and moisture in the air. This layer of oxides and hydroxides can prevent further contact with the fresh, reactive sodium metal underneath. By storing sodium in kerosene oil, which does not react with sodium, it is effectively isolated from the environment, ensuring its purity and preventing any potential hazards associated with its reactivity. - Equations for the Reactions:(i) Iron with Steam:
Iron reacts with steam (water vapor) to produce iron(II) oxide and hydrogen gas:
Fe(s) + H2O(g) → FeO(s) + H2(g)The iron reacts with water vapor to form iron oxide (FeO) and hydrogen gas (H2).
(ii) Calcium and Potassium with Water:
(a) Calcium with Water: Calcium reacts vigorously with water to form calcium hydroxide and hydrogen gas: Ca(s) + 2H2O(l) → Ca(OH)2(aq) + H2(g)
(b) Potassium with Water: Potassium reacts even more vigorously with water to produce potassium hydroxide and hydrogen gas: 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g)In both reactions, the alkaline earth metal calcium and alkali metal potassium react with water to form the respective metal hydroxides and hydrogen gas. - When dilute hydrochloric acid (HCl) is added to a reactive metal, hydrogen gas (H2) is produced. Here’s the chemical reaction when iron (Fe) reacts with dilute sulfuric acid (H2SO4): Fe(s) + H2SO4(aq) → FeSO4(aq) + H2(g)In this reaction, iron displaces hydrogen from sulfuric acid to form ferrous sulfate (FeSO4) and hydrogen gas (H2) is liberated.
- When zinc (Zn) is added to a solution of iron(II) sulfate (FeSO4), a displacement reaction occurs. You would observe that the zinc displaces iron from the iron(II) sulfate solution. Here’s the chemical reaction: Zn(s) + FeSO4(aq) → ZnSO4(aq) + Fe(s)In this reaction, zinc displaces iron from the solution, forming zinc sulfate (ZnSO4) and depositing iron metal. This type of reaction is known as a single displacement reaction, where one element replaces another in a compound.
Page No: 49

(ii) Formation of Na2O and MgO
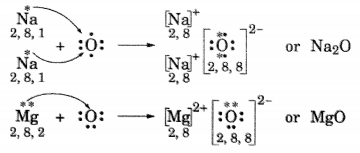
(iii) In Na2O, ions present are Na+ and O2-.
In MgO, ions present are Mg2+ and O2-.
2. Ionic compounds have strong electrostatic forces of attraction between the ions.
Therefore, it requires a lot of energy to overcome these forces. That is why ionic compounds have
high melting points

Leave a Reply