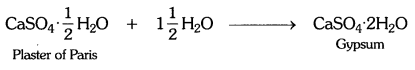NCERT Solutions for Class 10 Chapter 2 Acids Bases and Salts: This article provides NCERT Solutions for Class 10 Chapter 2 Acids and Bases. Having a thorough understanding of theories, adequate practice of reactions, equations, and formulas, as well as solving questions from NCERT Chemistry books, is essential for achieving a high score in Science in Class 10 board exams, Joint Examinations (JEE) and National Eligibility-cum-Entrance Test (NEET). Going through the stepwise solutions for each question is crucial for achieving a good score.
NCERT Class 10 Science Chapter 2 Notes for Acids, Bases and Salts are provided by the best Chemistry teachers in India. You will also find the step-wise explanation for every question in this article. Going through this step-by-step explanation will help you to understand how to solve the problems better. Continue reading to know NCERT solutions for class 10 science chapter 2 exercise and extra questions.

NCERT Solution for Class 10 Sciences Chapter 2 Acids, Bases and Salts
Before going into details about NCERT solutions for Class 10 Science Chapter 2 acids and bases, let us have a brief overview of topics and subtopics of NCERT solutions for Class 10 Science Chapter 2 activities:
- Acids and bases and salts
- Chemical properties of acids and bases
- What do all acids and bases have in common?
- How strong are acid or base solutions?
- More about salts
NCERT Solutions for class 10 science Chapter 2 Acids and bases PDF available for free download in Hindi Medium and English Medium for students of CBSE, Bihar, MP, Gujarat and UP Board students using NCERT books based on the updated CBSE syllabus 2022-23
NCERT Solutions for Class 10 Science Chapter 2 Textbook Questions
Question 1
A solution turns red litmus blue, its pH is likely to be
(a) 1
(b) 4
(c) 5
(d) 10
Answer:
(d) 10
Question 2
A solution reacts with crushed eggshells to give a gas that turns lime-water milky.
The solution contains
(a) NaCl
(b) HCl
(c) LiCl
(d) KCl
Answer:
(b) HCl
Question 3
10 mL of a solution of NaOH is found to be completely neutralized by 8 mL of a
given solution of HCl. If we take 20 mL of the same solution of NaOH, the amount
HCl solution (the same solution as before) required to neutralize it will be
(a) 4 mL
(b) 8 mL
(c) 12 mL
(d) 16 mL
Answer:
(d) 16 mL
Question 4
Which one of the following types of medicines is used for treating indigestion?
(a) Antibiotic
(b) Analgesic
(c) Antacid
(d) Antiseptic
Answer:
(c) Antacid
Question 5
Write word equations and then balanced equations for the reaction taking place when-
(a) dilute sulphuric acid reacts with zinc granules
(b) dilute hydrochloric acid reacts with magnesium ribbon
(c) dilute sulphuric acid reacts with aluminium powder
(d) dilute hydrochloric acid reacts with iron filing
Answer:
(a) Zinc + dilute sulphuric acid → Zinc sulphate + Hydrogen
Zn (s) + H2SO4 (aq) → ZnSO4 (aq) + H2 (g)
(b) Magnesium ribbon + dil. Hydrochloric acid → Magnesium chloride + Hydrogen
Mg (s) + 2 HCl (aq) → MgCl2 (aq) + H2 (g)
(c) Aluminium powder + Sulphuric acid > Aluminium sulphate + Hydrogen
2Al (s) + 3H2SO4 (aq) → Al2 (SO4)3 (aq) + 3H2 (g)
(d) Iron filings + Dilute hydrochloric acid > Ferric chloride + Hydrogen
2Fe (s) + 6HCl (aq) → 2FeCl3 (aq) + 3H2 (g)
Question 6
Compounds such as alcohol and glucose also contain hydrogen but are not categorized as acids. Describe an activity to prove it.
Answer:
Though compounds like alcohol and glucose contain hydrogen they do not ionise in the solution to produce H+ ions on passing current through them.
step1: Take solutions of alcohol and glucose.
Step2: Attach two nails to a cork
Step3: Put the cork into a 100 ml beaker
Step4: Attach the nails to the terminals of the 6-volt battery
Step5: Use a light bulb and a switch to
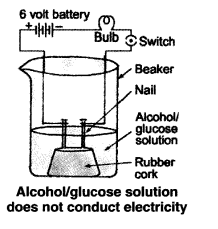
Step6: Now add the alcohol to the beaker and turn on the current
Setp7: The bulb does not light up
Step8: Turn on the glucose
Step9: The bulb also does not light up in this case
Step10: There are no H+ ions in the solution
Step11: This indicates that alcohol and glucose are not acids
Question 7
Why does distilled water not conduct electricity, whereas rainwater does?
Answer:
The reason why distilled water doesn’t conduct electricity is because it doesn’t contain any ions (e.g., acids, bases, salts). On the other hand, rainwater does. As it falls to the ground from the atmosphere, it breaks down an acidic gas, carbon dioxide, from the air to form carbonic acid. Carbonic acid, on the other hand, provides hydrogen ions (H+ (aq)) and carbonate (CO(aq) 32) ions to the rainwater. So, because carbonic acid provides ions to the rainwater, it conducts electricity.
Question 8
Why do acids not show acidic behaviour in the absence of water?
Answer:
Acids are acidic because hydrogen ions (aq) are present in them. Acids only form hydrogen ions when they’re in contact with water. Without water, an acid won’t form hydrogen ions and won’t be acidic.
Question 9
Five solutions A, B, C, D and E when tested with universal indicator showed pH as 4, 1, 11, 7 and 9 respectively. Which solution is
(a) Neutral
(b) Strongly alkaline
(c) Strongly acidic
(d) Weakly acidic
(e) Weakly alkaline
Arrange the pH in increasing order of hydrogen ion concentration.
Answer:
(a) D
(b) C
(c) B
(d) A
(e) E
Increasing the order of hydrogen ion concentration
C < E < D < A < B
Question 10
Equal lengths of magnesium ribbons are taken in test tubes A and B. Hydrochloric acid (HCl) is added to test tube A, while acetic acid (CH3COOH) is added to test tube B. In which test tube will the fizzing occur more vigorously and why?
Answer:
Test tube A will fizz more. Acetic acid is a weak acid, while hydrochloric acid is a strong acid. Due to the strong acid nature of hydrochloric acid, the solution with hydrochloric acid contains many more hydrogen ions than acetic acid. Therefore, the fizzing of test tube A (which contains hydrochloric acid) will be more intense.
The fizzing is caused by the formation of hydrogen gas. Hydrogen gas is formed when acid reacts with the magnesium metal of the magnesium ribbon.
Question 11
fresh milk has a pH of 6. How do you think the pH will change as it turns into curd? Explain your answer.
Answer:
Lactic acid reduces the pH value of the milk. When lactic acid is present in the milk, the pH value drops below 6 as the milk turns into a curd.
Question 12
A milkman adds a very small amount of baking soda to fresh milk.
(a) Why does he shift the pH of the fresh milk from 6 to slightly alkaline?
(b) Why does this milk take a long time to set as curd?
Answer:
(a) Because milk contains lactic acid, it is slightly alkaline, so it may not sour as easily.
(b) It takes longer for the alkaline milk to settle into the curd, as the lactic acid must first neutralize the alkali in the milk.
Question 13
Plaster of Paris should be stored in a moisture-proof container. Explain why?
Answer:
Plaster of Paris should be stored in a moisture-proof container because the presence of moisture can cause slow setting of plaster of Paris by bringing about its hydration. This will make the plaster of Paris useless after some time.
Question 14
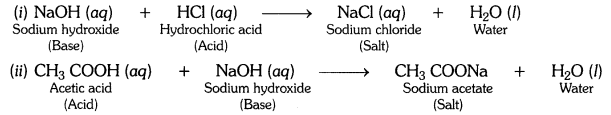
What is a neutralization reaction? Give two examples.
Answer:
The reaction between an acid and a base to form salt and water is called a neutralization reaction.
Question 15
Give two important uses of washing soda and baking soda.
Answer:
Uses of washing soda :
(i) Washing soda is used in glass, soap and paper industries.
(ii) It is used for removing the permanent hardness of water.
Uses of baking soda :
(i) Baking soda is used as an antacid in medicines to remove acidity in the stomach.
(ii) Baking soda is used for making baking powder (used in making cakes, bread, etc.).
| Board | CBSE, UP, GSEB |
| Class | 10th |
| TextBook | NCERT |
| Chapter | 2 |
| Chapter Name | Acids, Base and Salt |
NCERT Solutions for Class 10 Sciences Chapter 2 Acids, Base and Salt
Page Number: 18
Question 1
You have been provided with three test tubes. One of them contains distilled water and the other two contain an acidic solution and a basic solution, respectively. If you are given only red litmus paper, how will you identify the contents of each test tube?
Answer:
(i) Put the red litmus paper in all the test tubes, turn by turn. The solution which turns red litmus to blue will be a basic solution. The blue litmus paper formed here can now be used to test the acidic solution.
(ii) Put the blue litmus paper obtained above in the remaining two test tubes, turn by turn. The solution which turns the blue litmus paper to red will be the acidic solution.
(iii) The solution which has no effect on any litmus paper will be neutral and hence it will be distilled water.
Question 1
Why should curd and sour substances not be kept in brass and copper vessels?
Answer:
Curd and sour substances should not be kept in brass and copper vessels because these and other sour food stuffs contain acids which can react with the metal of the vessel to form poisonous metal compounds which can cause food poisoning and affect our health adversely.
Question 2
Which gas is usually liberated when an acid reacts with a metal? Illustrate with an example. How will you test for the presence of this gas?
Answer:
(i) Hydrogen (H2) gas is liberated when an acid reacts with a metal.
(ii) Illustration: Set up the apparatus as shown in the given figure. Take some zinc granules in the test tube. Add about 5 mL dilute hydrochloric acid slowly. Soon the reaction between zinc and hydrochloric acid starts and hydrogen gas is evolved.
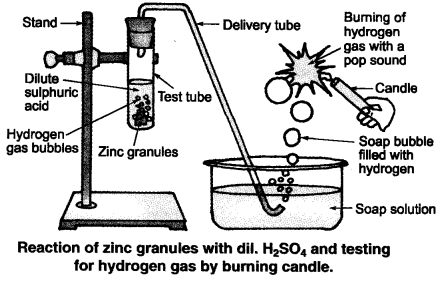
(iii) Test for H2 gas :
H2 gas is not soluble in water. When passed through soap solution, it gets trapped into bubbles.
Bring a burning candle near the soap bubble filled with gas. The soap bubble bursts and hydrogen gas burns with a pop sound.
Question 3
Metal compound A reacts with dilute hydrochloric acid to produce effervescence. The gas evolved extinguishes a burning candle. Write a balanced chemical equation for the reaction if one of the compounds formed is calcium chloride.
Answer:
As the end product is calcium chloride and the gas formed is carbon dioxide, the metal compound A must be calcium carbonate. Therefore, the reaction between calcium carbonate and hydrochloric acid is
Question 1
Why do HCl, HNO3, etc show acidic characters in aqueous solutions while solutions of compounds like alcohol and glucose do not show acidic character?
Answer:
H+ ions in aqueous solution are responsible for acidic character. HCl, HNO3, etc. give H+ ions in water while alcohol and glucose do not give H+ ions in water. Therefore, alcohol and glucose do not show an acidic character.
Question 2
Why does an aqueous solution of an acid conduct electricity?
Answer:
The aqueous solution of an acid conducts electricity due to the presence of charged particles called ions in it.
Question 3
Why does dry HCl gas not change the colour of the dry litmus paper?
Answer:
Dry HCl gas does not give H+ ions and therefore does not change the colour of dry litmus paper.
Question 1
You have two solutions A and B. The pH of solution A is 6 and the pH of solution B is 8. Which solution has more hydrogen ion concentration? Which of these is acidic and which one is basic?
Answer:
A pH value of less than 7 indicates an acidic solution, while greater than 7 indicates a basic solution. Since solution A has more hydrogen ion concentration, solution A is acidic and solution B is basic.
Question 2
What effect does the concentration of H+ (aq) ions have on the nature of the solution?
Answer:
The higher the concentration of H+ ions, the higher the acidic nature of the solution.
Question 3
Do basic solutions also have H+ (aq) ions? If yes, then why are these basic?
Answer:
Basic solutions have H+ (aq) ions. But these are far less in number than OH– ions that are responsible for their basic nature.
Question 4
Under what soil condition do you think a farmer would treat the soil of his fields with quick lime (calcium oxide) slaked lime (calcium hydroxide) or chalk (calcium carbonate)?
Answer:
If the soil is too acidic (having low pH) then it is treated with materials like quick lime (calcium oxide) slaked lime (calcium hydroxide) or chalk (calcium carbonate).
Question 1
What is the common name of the compound CaOCl2?
Answer:
Bleaching powder.
Question 2
Name the substance which on treatment with chlorine yields bleaching powder.
Answer:
Slaked lime Ca (OH)2.
Question 3
Name the sodium compound which is used for softening hard water.
Answer:
Sodium carbonate.
Question 4
What will happen if a solution of sodium hydrogen carbonate is heated? Give the equation of the reaction involved.
Answer:
The solution of sodium hydrogen carbonate on heating gives sodium carbonate and carbon dioxide gas.
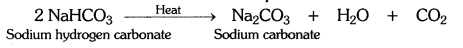
Question 5
Write an equation to show the reaction between the plaster of Paris and water.
Answer: